Why is manganese dioxide surface area important in technology? Scientists find that a bigger surface area helps batteries and catalysts work better. The table below shows how different kinds of manganese dioxide with different surface areas can change how batteries work. It also shows the specific surface area measured by BET.
MnO2 Polymorph | BET Surface Area (m²/g) | Initial Discharge Capacity (mAh/g at 0.1 A/g) |
|---|---|---|
α-MnO2 | (reported) | 230.5 |
β-MnO2 | (reported) | 188.74 |
δ-MnO2 | (reported) | 263.30 |
When manganese dioxide has a bigger surface area, it works better in batteries and as a catalyst. This is because there are more places for reactions to happen.
The BET method uses nitrogen gas at very low temperatures. It measures the specific surface area. This helps scientists learn about manganese dioxide. It also helps them make it work better.
Changing the crystal form, nanostructure, and how manganese dioxide is made can make its surface area bigger. This makes it work better for storing energy and cleaning up pollution.
BET and Manganese Dioxide Surface Area
BET Method Overview
The BET method helps scientists find the specific surface area of manganese dioxide. It is based on Langmuir’s idea, where one layer of gas molecules sticks to a surface. BET lets more than one layer of gas molecules stick to the surface. The method says each layer needs the same energy. It also says the gas molecules do not affect each other. Scientists use the BET equation to link the amount of gas to the pressure. This helps them figure out the specific surface area. Nitrogen gas is used for this test at very cold temperatures, like 77 K.
The BET method uses nitrogen gas to cover the manganese dioxide surface at very cold temperatures. This shows how much gas the surface can hold. It tells scientists about the high surface area and the holes in the material.
Specific Surface Area Measurement
Manganese dioxide can have many different specific surface area values. For example, electrolytic manganese dioxide can have values from 12.1 to 52.0 m²/g. Manganese dioxide made in labs can have much higher values, even up to 525 m²/g. The way it is made changes the value. Scientists use nitrogen gas at -196°C to make graphs called isotherms. These graphs show how much nitrogen the manganese dioxide can hold at different pressures. The graphs help find the specific surface area and the size of the holes. The specific surface area changes how well manganese dioxide works in batteries and as a catalyst. A high surface area gives more places for reactions. This makes batteries work better.
Crystal Forms and Nanostructures
Manganese dioxide has different crystal forms, like α-MnO2, β-MnO2, γ-MnO2, and δ-MnO2. Each form has its own specific surface area and works differently in batteries. The table below shows how the forms compare:
Crystal Form | Specific Surface Area (m²/g) | Average Particle Size (nm) | Pore Size (nm) | Electrochemical Impact |
|---|---|---|---|---|
γ-MnO2 | ~154 | ~39 | ~14.1 | Largest surface area, best for charge transfer and redox reactions |
δ-MnO2 | ~149 | ~40 | ~9.2 | Large surface area, rapid redox reactions, best capacitive performance |
β-MnO2 | ~44 | ~136 | ~15 | Smaller surface area, mainly capacitive behavior |
α-MnO2 | ~52 | ~114 | ~9.2 | Moderate surface area, lower capacity |
ε-MnO2 | ~39 | ~153 | ~5 | Smallest surface area, poor performance |
Nanostructures are also important. Nanoflowers and nanorods are two common shapes. Nanoflowers have a higher specific surface area than nanorods. They also work better in batteries. The 3D shape of nanoflowers lets them touch more of the electrolyte. It also gives ions and electrons shorter paths to move.
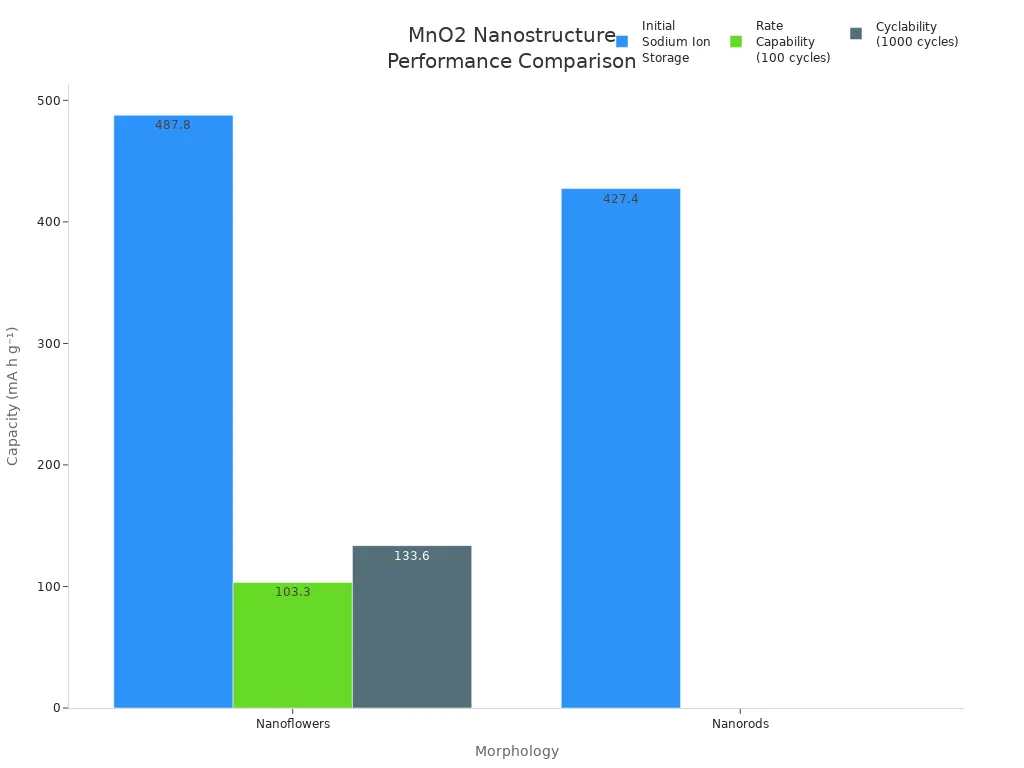
The surface area of manganese dioxide depends on the crystal form and the nanostructure. A high specific surface area helps manganese dioxide work better in batteries and catalysts.
Surface Area and Electrochemical Performance

Role in Batteries and Catalysis
Manganese dioxide is important in batteries and catalysis because of its specific surface area. When the surface area is high, more ions can reach the surface. This gives more spots for surface redox reactions. If manganese dioxide has a surface area above 70 m²/g, batteries can hold about 250 mAh/g. This means batteries work better when the electrochemically active surface area is bigger. The boost comes from reactions on the surface, not just inside the material. During battery use, a non-crystalline layer forms on the surface. This layer helps with redox reactions and makes the electroactive surface area larger.
In catalysis, manganese dioxide with a high surface area shows more active sites. Two-dimensional nanosheets have a large specific surface area. This shape lets more reactants, like protons and OH- ions, stick to the surface. This makes electron and charge transfer faster, so the catalyst works better. Scientists can add elements like phosphorus or iron to the surface. This makes the electrochemically active surface area even bigger. These changes lower the energy needed for reactions. This makes manganese dioxide great for water splitting and other catalytic jobs.
The crystal form and shape of manganese dioxide change its surface area and how well it works. For example, δ-MnO2 has layers that show many edge sites. These sites are very good for catalysis. When scientists put manganese dioxide on stable bases, they get more active sites and better stability. This helps manganese dioxide work better in real uses, like breaking down hydrogen peroxide or storing energy.
Adsorption Properties
The specific surface area of manganese dioxide also affects how well it can adsorb ions and pollutants. A high surface area gives more places for ions or molecules to stick. For example, MnO2@SiO2 colloids with a large electrochemically active surface area can hold more Mn2+ ions. If the particles stick together, the surface area goes down and so does adsorption. At low concentrations, the adsorption can be very high. This shows that a big surface area is important for adsorption.
Other studies show that spreading manganese dioxide on a support increases the surface area. This also increases how much heavy metal, like Pb(II), it can adsorb. If the particles group together, there are fewer spots for adsorption and the capacity drops. Nanorods with a high specific surface area can adsorb more organic pollutants, like methylene blue. The shape and size of the particles matter a lot for cleaning up pollutants.
The shape of manganese dioxide changes how it adsorbs things. For example, mesoporous fibrillar manganese dioxide made by hydrothermal synthesis has a surface area of 378.1 m²/g and can adsorb 99.6% of 2,4-dinitrophenol. Short rod-like manganese dioxide has a smaller surface area and adsorbs less. The mesoporous structure gives more space for molecules to enter and react. This makes the electrochemically active surface area bigger and improves adsorption.
Thin Films of Electrodeposited Manganese Dioxide
Thin films of electrodeposited manganese dioxide are used in supercapacitor electrodes and energy storage. Scientists can control the surface area and shape of these films by changing how they make them. Short electrodeposition times make thin, porous films with high surface area and good performance. These films let ions move fast, which helps with redox reactions and makes the electrochemically active surface area bigger.
If the deposition time is longer, the films get thicker and denser. This lowers the surface area and makes it harder for ions and electrons to move. The performance drops because redox reactions cannot happen as easily. Scanning electron microscopy shows that short deposition times make nanobelt shapes. These have a high surface area and help the electrochemically active surface area. These shapes are great for supercapacitor electrodes, where fast charging is needed.
Different electrodeposition methods, like potentiostatic, galvanostatic, and pulsed, make films with different nanostructures. For example, using potentiostatic and cyclic voltammetric electrodeposition can make γ-MnO2 films with carambola-like nanoflakes. These films have a high surface area and work very well, with high capacitance and good stability. The way scientists deposit the films and how long they do it are important for making the best supercapacitor electrodes.
Crystallinity also changes how manganese dioxide works. When crystallinity goes up, the surface area and number of active sites go up too. For example, making γ-MnO2 more crystalline changes its shape from pine-needle-like to daisy-like. This increases double-layer capacitance and makes the electrochemically active surface area bigger. This helps store more energy and makes electrodes more stable for capacitors.
Factors Affecting BET Results
Sample Preparation
Getting manganese dioxide ready is very important for BET tests. Scientists must dry and degas the samples first. This step takes away water and gases stuck on the surface. If these are not gone, nitrogen cannot reach every spot. Then, the specific surface area will look too small. For example, scientists often dry and degas manganese dioxide at 250°C for many hours. This heat gets rid of water but does not hurt the structure. Too much heat can change the shape or break down the manganese dioxide. It can also make the specific surface area smaller. Scientists use tools like TGA and DSC to pick the best temperature. The goal is to keep the surface clean and the sample the same for good results.
Tip: Make sure the manganese dioxide is dry and has no water on the surface before BET testing. This helps you get the real specific surface area.
Morphology and Stability
The shape and size of manganese dioxide particles matter a lot. They change the specific surface area and how well the material works in batteries. Nanoparticles have a much bigger specific surface area than large pieces. This means more places for reactions and better energy storage. Nanostructured manganese dioxide also lets ions move faster. This is good for electrodes. But tiny particles need protection to stay stable. Coatings and careful control help keep the specific surface area high. They also help the electrochemically active surface area stay steady during use.
Heating can change the specific surface area of manganese dioxide. Heating at 400°C can make more pores and raise the specific surface area. But if the heat goes up to 600°C, the manganese dioxide can change form and lose oxygen. Then, the specific surface area drops. This also makes the electrochemically active surface area smaller, which is not good for batteries.
Scientists have some problems when they measure the specific surface area. Sometimes, BET results do not match the real electrochemically active surface area. The nitrogen in BET tests is not like the ions in batteries. Also, BET works best for powders, not thin films. To get the best answers, scientists use other tests with BET. They always think about the shape, how well it conducts, and how stable the manganese dioxide is.
Making the surface area of manganese dioxide bigger helps it work better in batteries and as a catalyst. BET analysis lets scientists check different ways to make manganese dioxide. This helps them pick the best one. If manganese dioxide has layers or special doping, it gets a higher surface area and works better.
The hard template method makes manganese dioxide with a big surface area.
BET and TEM show how making methods change manganese dioxide.
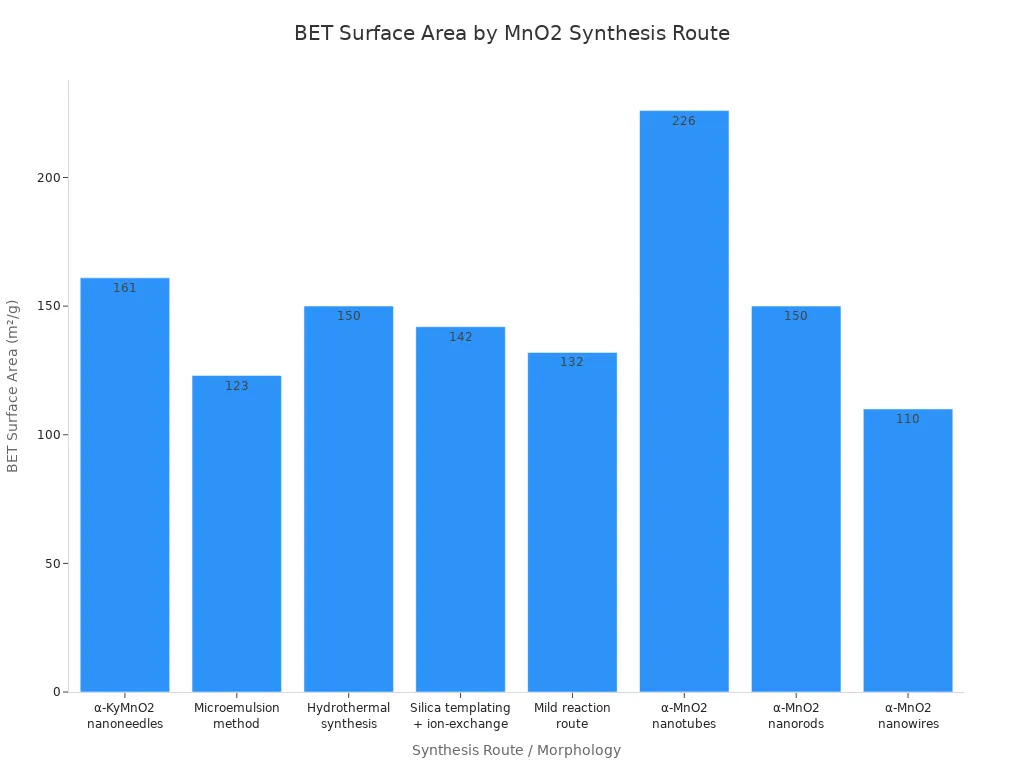
Careful measuring and picking the right way to make manganese dioxide helps scientists design it for better batteries and catalysts.
FAQ
What does BET stand for in surface area analysis?
BET means Brunauer–Emmett–Teller. Scientists use this method to find the specific surface area of things like manganese dioxide.
Why do scientists use nitrogen gas in BET analysis?
Nitrogen gas covers the manganese dioxide surface at very cold temperatures. This lets scientists see how much surface area the material has.
Can BET analysis measure thin films of manganese dioxide?
BET is best for powders, not thin films. Thin films might not give good results because nitrogen gas cannot reach all parts of the film.
Related Posts

I am Edward lee, founder of manganesesupply( btlnewmaterial) , with more than 15 years experience in manganese products R&D and international sales, I helped more than 50+ corporates and am devoted to providing solutions to clients business.





