Manganese dioxide pharmaceutical materials possess unique chemical properties that enhance the development of new medicines. The nanosheet structure of manganese dioxide pharmaceutical compounds provides a large surface area, which is beneficial for biosensing and drug delivery applications. Its redox activity allows healthcare professionals to precisely control drug release and enables imaging techniques that can be activated on demand. These features improve both imaging and therapy, making it easier to detect diseases and provide effective treatment.
Chemical Property | Description | Medical Application |
|---|---|---|
Nanosheet structure | Large surface area | Biosensing, drug delivery |
Redox activity | Works with glutathione | Controlled release, imaging |
Mn2+ degradation | Paramagnetic ions | MRI imaging |
Biocompatibility | Many forms | Used in many medical ways |
Manganese dioxide nanosheets and nanoparticles help deliver drugs better. They release medicine only at the right spot. This lowers side effects and makes treatment work well.
Its special features help in cancer therapy and imaging. Tumors are easier to find and treat. This gives better accuracy and less risk.
Safety depends on how much and what type is used. Careful tests and rules keep manganese dioxide safe for medical use. This helps future clinical uses too.
Functions
Catalytic Activity
Manganese dioxide acts as a helper in making medicines. Scientists use it to make chemical reactions go faster. This helps them make important parts of drugs. For example, manganese dioxide on magnetic nanoparticles changes secondary alcohols into ketones. This step is needed to make drugs like tamoxifen and ibuprofen. The catalyst works well even after being used many times. It can be taken out easily, so there is less waste and fewer problems with mixing. Manganese–porphyrin complexes on magnetic carbon-coated nanoparticles help make chiral epoxides. These are important for drugs like propranolol, which is a β-blocker. Magnetic manganese oxides help with C–H amination. This is useful for making antiviral drugs like oseltamivir. These systems work well, can be used again, and are better for the environment.
Catalyst System | Reaction Type | Pharmaceutical Application | Effectiveness |
|---|---|---|---|
MnO2/Fe3O4@SiO2 | Alcohol oxidation | Tamoxifen, ibuprofen | |
Mn–porphyrin/carbon | Asymmetric epoxidation | Propranolol | High selectivity, easy purification |
Magnetic Mn oxides | C–H amination | Oseltamivir | Cost-effective, low contamination |
Oxidative Properties
Manganese dioxide has strong oxidative properties that help in therapy and drug delivery. Its nanoenzymes act like natural enzymes and break down hydrogen peroxide. This makes oxygen and helps lower stress in diseases like cancer and inflammation. Scientists made manganese dioxide nanoparticles that can go into tissues and protect them from harm. These particles also work with glutathione to change the tumor area and help drugs stay longer. In drug delivery, manganese dioxide lets drugs out when certain things happen, like low pH or lots of hydrogen peroxide. This way, drugs work better and have fewer side effects. Manganese dioxide also helps smart delivery systems send drugs to the right place at the right time.
MnO2 nanoparticles break down hydrogen peroxide and lower stress.
They make oxygen in tumors, which helps drug delivery and therapy.
MnO2 lets drugs out when tumor conditions are right.
These features make manganese dioxide useful for drug delivery and imaging.
Manganese Dioxide Pharmaceutical Applications
Antipyretic Formulations
Manganese dioxide pharmaceutical compounds are important in fever medicine. These biomedical applications help lower fever by breaking down bad substances. Scientists use manganese dioxide pharmaceutical materials to make fever drugs work better. The special oxidative properties of manganese dioxide pharmaceutical agents help control how drugs are released. This control gives more steady and expected results when treating fever. These uses also make fever drugs safer for people. Many biomedical applications now use manganese dioxide pharmaceutical ingredients to deliver drugs well and cause fewer side effects. The big surface area of manganese dioxide pharmaceutical nanosheets helps active ingredients get absorbed quickly. This feature makes these biomedical applications useful in hospitals and at home.
Biosensing
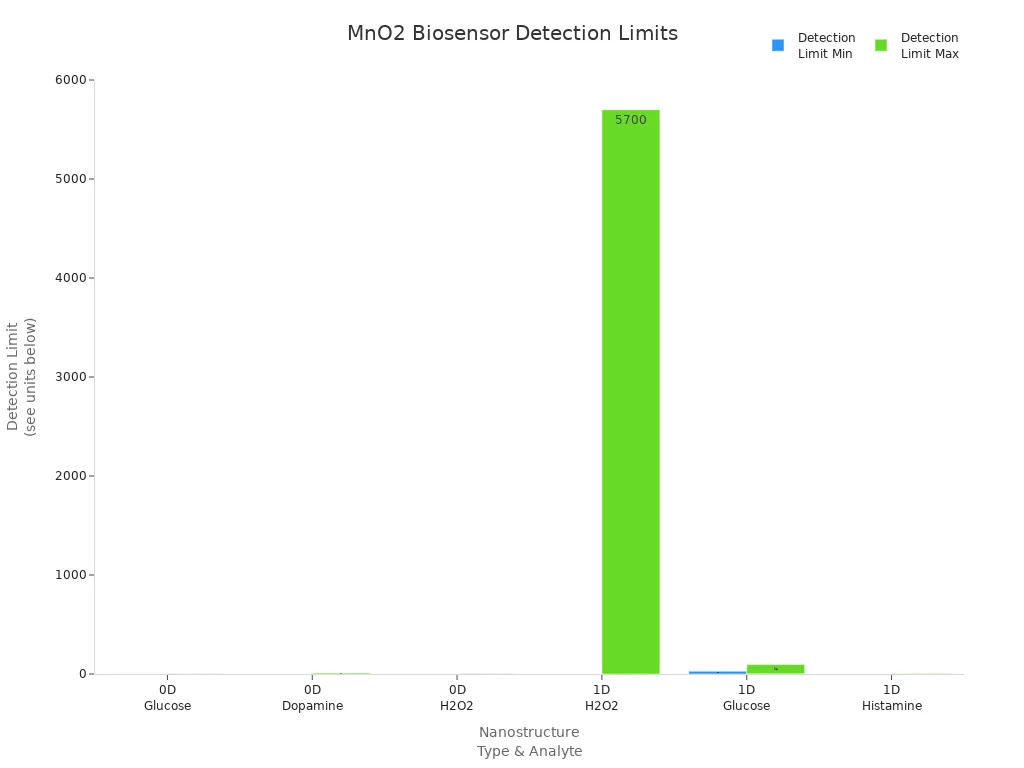
Biosensing is a very advanced use for manganese dioxide pharmaceutical materials. Researchers make biosensors with different types of manganese dioxide pharmaceutical nanostructures. These biosensors can find many things in blood, serum, food, and other samples. The table below shows some biomedical uses of manganese dioxide pharmaceutical biosensors, their detection limits, and what they look for:
MnO2 Nanostructure Type | Biosensor Configuration | Sample Matrix | Target Analyte | Detection Limit Range | Sensing Technique |
|---|---|---|---|---|---|
0-Dimensional (Nanoparticles) | MnO2 nanoparticles on gold nanorods composite | Honey | Glucose | 0.1 to 1.4 mM | Electrochemical |
0-Dimensional (Nanoparticles) | MnO2 nanoparticles with polythiophene on glassy carbon electrode | Human serum | Dopamine | 0.04 to 9.0 μM | Electrochemical |
0-Dimensional (Nanoparticles) | MnO2 nanoparticles on tantalum electrode | Milk | Hydrogen peroxide (H2O2) | 1 to 2 μM | Cyclic Voltammetry and Amperometry |
1-Dimensional (Nanotubes, Nanowires) | MnO2 nanotubes with Ag@C shell nanocomposites | Toothpaste | Hydrogen peroxide (H2O2) | 0.5 μM to 5.7 mM | Electrochemical |
1-Dimensional (Nanowires) | M13 bacteriophage-templated MnO2 nanowires | Human serum, peach juice | Glucose | 5 μM to 2 mM | Electrochemical |
1-Dimensional (Nanorods) | Au/MnO2 nanodendrites on screen-printed carbon electrode | Blood plasma | Histamine | 0.3 to 5.1 μM | Amperometry |
1-Dimensional (Nanorods) | MnO2 nanorods on hollow carbon spheres | Blood sample | Glucose | 28 to 93 μg/mL | Cyclic Voltammetry and Chrono |
Manganese dioxide pharmaceutical nanosheets help make biosensors that use two ways to find things. These biomedical applications use both glowing and color changes to find bacteria like Staphylococcus aureus in food. The biosensor can find as few as 9 CFU/mL with glowing and 22 CFU/mL with color change. This strong sensitivity makes manganese dioxide pharmaceutical biosensors good for food safety and medical tests. Biomedical applications keep growing as researchers find new ways to use manganese dioxide pharmaceutical materials for fast and correct detection.
Drug Delivery
Manganese Dioxide Nanosheets
Manganese dioxide nanosheets are important for drug delivery. Their special shape gives them a big surface area. This lets them hold more drug molecules than regular carriers. Scientists use these nanosheets to help drugs reach the right spot in the body. The thin, flat shape helps them move through tissues and get into cells easily. Researchers learned that manganese dioxide nanosheets can work as smart nanocarriers. They can grab single-stranded DNA, which helps them get into cells. In the body, cancer cells have lots of glutathione. This makes the manganese dioxide nanosheets break down. When this happens, the drug is released at the right time and place. The nanosheets also help break down hydrogen peroxide. This makes oxygen, which helps treat tumors better.
Note: Manganese dioxide nanosheets can cover other nanoparticles. This adds new features like imaging and sensing to drug delivery.
The table below shows how manganese dioxide nanosheets and nanoparticles carry drugs in biomedical applications:
Mechanism | Description |
|---|---|
Nanosheets grab DNA, which helps them get into cells. | |
Glutathione in cancer cells breaks down MnO2, releasing the drug and keeping DNAzyme working. | |
Nanosheets break down hydrogen peroxide, make oxygen, and help cancer therapy. | |
Gatekeeper function | Nanoshells stop drugs from leaking out too soon and let them out when they break down. |
GSH-responsiveness for targeting | High glutathione in tumors makes drugs come out, so there are fewer side effects. |
Coating on other nanoplatforms | Nanosheets can cover other nanoparticles for smart delivery and imaging. |
Manganese dioxide nanosheets also help change the tumor area. They give off oxygen and manganese ions. This wakes up immune cells and lowers bad molecules. These changes help drugs stay in the tumor longer and work better. Biomedical uses with these nanosheets show drugs last longer and go deeper into tissues. This makes them helpful for cancer treatment.
Controlled Release
Controlled drug delivery is a big benefit of manganese dioxide nanosheets and nanoparticles. These carriers react to things inside the body, like low pH or lots of glutathione, which are found in cancer cells. When these things are present, the carriers break down and let the drug out slowly. This way, the drug goes right where it is needed and causes fewer side effects.
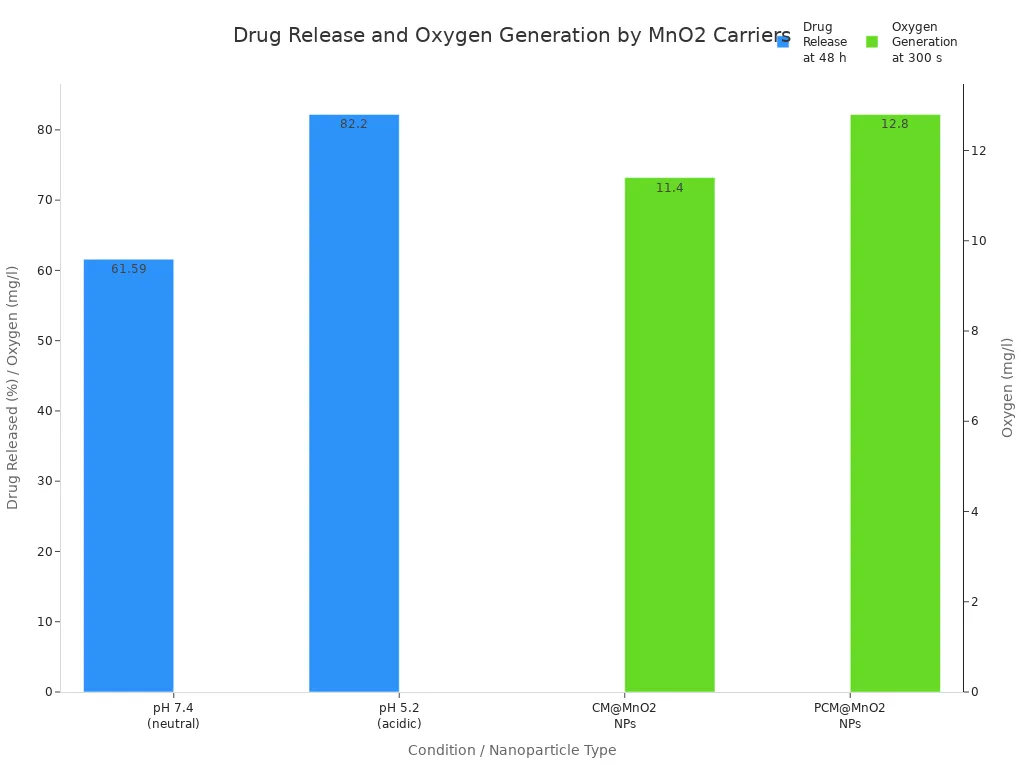
The next table shows how manganese dioxide carriers control drug release in different situations:
Condition | Drug Released (%) | Time | Notes |
|---|---|---|---|
pH 7.4 (neutral) | 48 h | Methotrexate comes out of PCM@MnO2 nanoparticles | |
pH 5.2 (acidic, M1 macrophages) | 82.2 ± 1.3 | 48 h | More drug comes out because of the acidic pH |
Dissolved oxygen (CM@MnO2 NPs) | 11.4 mg/l | 300 s | Oxygen is made when MnO2 breaks down H2O2 |
Dissolved oxygen (PCM@MnO2 NPs) | 12.8 mg/l | 300 s | More oxygen is made with a PDA coating |
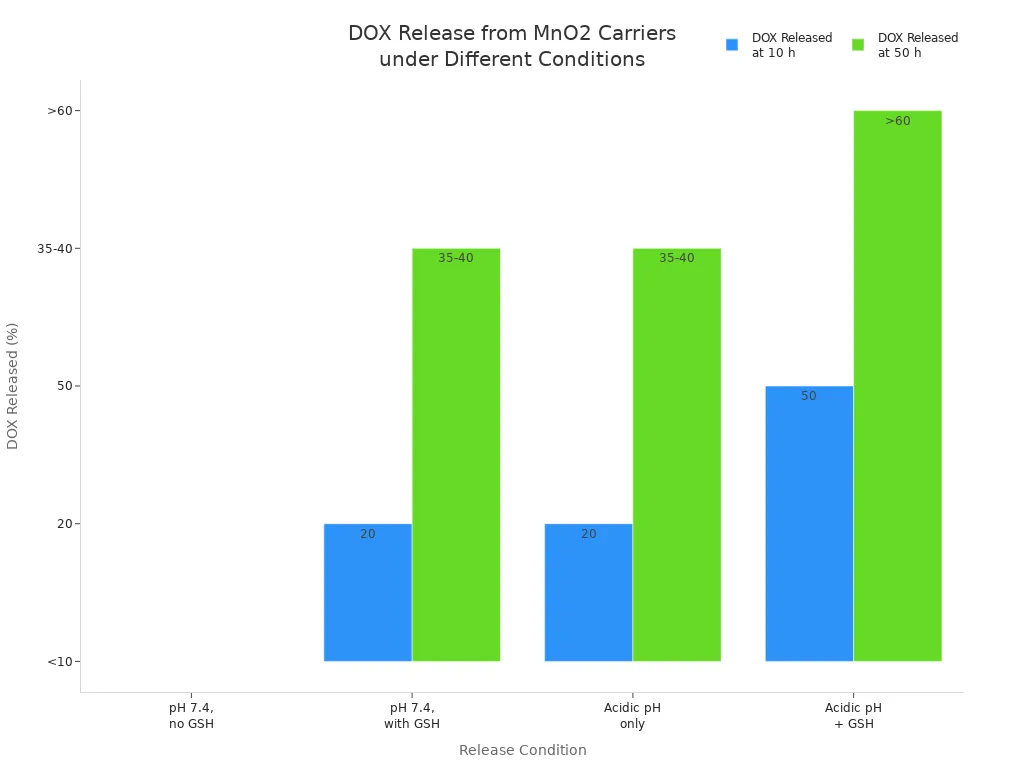
Another study checked how doxorubicin (DOX), a cancer drug, comes out of manganese dioxide nanocarriers. The results showed that both pH and glutathione levels matter:
Condition | DOX Released (%) | Time | Description |
|---|---|---|---|
pH 7.4, no GSH | 50 h | Very little drug comes out; carrier stays together | |
pH 7.4, with GSH (5-10 mM) | ~20 (10 h), up to 35-40 (50 h) | 50 h | Glutathione helps break down the carrier and lets some drug out |
Acidic pH only | ~20 (10 h), up to 35-40 (50 h) | 50 h | Acidic pH also helps break down the carrier and lets some drug out |
Acidic pH + GSH | ~50 (10 h), >60 (50 h) | 50 h | Both together break down the carrier the most and let out the most drug |
These results show that manganese dioxide nanosheets and nanoparticles can control when drugs are released. They only let the drug out when they reach the right spot, like a cancer cell. This keeps healthy tissues safe and helps cancer treatments work better.
Manganese dioxide nanocarriers also help change the tumor area. They give off oxygen and manganese ions, which wake up immune cells and lower bad molecules. These changes make it harder for tumors to fight treatment and help drugs stay in the tumor longer. Studies show these uses can slow tumor growth and help animals live longer. By helping drugs last longer and go deeper, manganese dioxide nanosheets make cancer treatment better.
Tip: Using manganese dioxide nanosheets for controlled drug delivery can mix therapy and imaging in one system. This makes them strong tools for modern biomedical uses.
Imaging
MRI Contrast
Manganese dioxide helps doctors see inside the body. Scientists use it in MRI scans as a contrast agent. When it goes into the body, it meets things in tumors like glutathione. This makes manganese dioxide change into Mn2+ ions. These ions make MRI pictures brighter. Doctors can spot tumors more easily. Manganese dioxide agents stay “off” in healthy areas. They turn “on” only in tumor spots. This lowers background noise in the images. It helps doctors find cancer more accurately. The strong MRI signals from Mn2+ ions make manganese dioxide very useful for imaging and theranostics.
Tumor Diagnosis
Manganese dioxide nanoparticles help doctors find tumors with high accuracy. These particles react to the tumor area, which has lots of glutathione and low pH. This means they only work in tumors, so imaging is more exact. The table below shows how manganese dioxide helps doctors diagnose tumors better:
Mechanism/Feature | Description | Impact on Tumor Diagnosis Accuracy |
|---|---|---|
Tumor Microenvironment Responsiveness | Reacts with high glutathione and acidic pH in tumors | Improves imaging specificity |
GSH Reduction Reaction | Produces Mn2+ ions for MRI | Increases signal-to-noise ratio |
Dual-Mode Imaging Capability | Combines MRI with fluorescence | Enhances diagnostic accuracy |
Activatable Contrast Agent | “Off” in normal tissue, “on” in tumors | Reduces false positives |
Enhanced Relaxation Rates | Stronger MRI signals at tumor sites | Aids precise localization |
Nanoparticle Engineering | Targets tumor cells | Improves sensitivity and specificity |
Theranostic uses get better with these features. Manganese dioxide lets doctors do imaging and therapy together. This helps cancer care in many ways. Researchers keep looking for new theranostic uses. Manganese dioxide is now a key material for diagnostics and theranostics.
Therapeutic Uses
Anti-Cancer
Manganese dioxide helps treat cancer in many ways. It makes targeted treatments work better. Scientists found that manganese dioxide nanomaterials help fix low oxygen in tumors. Low oxygen makes cancer hard to treat. These materials act like enzymes. They break down hydrogen peroxide in tumors and make oxygen. This helps photodynamic therapy and radiotherapy work better. Cancer cells become easier to kill.
Manganese dioxide is used in theranostics to increase stress in cancer cells. It uses up glutathione and makes more reactive oxygen species. This causes more harm to cancer cells. Nanostructured manganese dioxide, like nanoflowers, works faster and helps deliver drugs to the right spot. These uses let drugs come out only in acidic or hydrogen peroxide-rich tumor areas.
Manganese dioxide also helps the immune system fight cancer. It helps dendritic cells grow up. It changes macrophages to a type that fights cancer. It wakes up CD8+ T cells and natural killer cells. These changes help the body attack cancer better. In theranostic systems, manganese dioxide helps with both imaging and therapy. This makes it easier to find and treat cancer.
Scientists mix manganese dioxide with other drugs and photosensitizers for photothermal therapy. When lasers shine on these mixes, they make heat and oxygen. This helps stop drug resistance and slows tumor growth. Manganese dioxide does many jobs in cancer therapy and theranostics.
Tip: Manganese dioxide nanoplatforms can help wounds heal. They change the tumor area and boost the immune system.
Anti-Bacterial
Manganese dioxide also kills bacteria well. Studies show manganese dioxide nanosheets kill over 99% of Salmonella at certain amounts. It works by making reactive oxygen species. These break open bacteria and make them leak. This kills the bacteria.
Adding silver to manganese dioxide makes it even stronger. These mixes stop bad bacteria like Pseudomonas aeruginosa and Staphylococcus aureus. The mix of manganese dioxide and silver is good for fighting infections in hospitals.
Scientists keep studying manganese dioxide for healing and infection control. It makes reactive oxygen species and breaks up bacteria. This helps with cancer and infection care. Manganese dioxide’s many uses help with drug delivery, cancer treatment, and killing bacteria.
Safety and Toxicity
Biocompatibility
Scientists have tested manganese dioxide in many medicine formulas. Most tests show manganese dioxide hydrogels and nanoreactors are safe for cells. For example, L929 rat fibroblast cells stayed healthy with some manganese dioxide nanoparticles. Some types, like commercial grade, were less harmful than others. Adding manganese dioxide to hydrogels with methacrylated gellan gum and hyaluronic acid helped control drug release and made them less toxic. These hydrogels also helped cells grow, especially with less hyaluronic acid. Sometimes, manganese dioxide nanoparticles removed reactive oxygen species and worked as MRI contrast agents. But high amounts of manganese dioxide alone can be harmful. Using polymers makes them safer and work better. A new terpolymer-lipid formula with manganese dioxide nanoparticles gave strong MRI signals and was safe at much higher doses than those used for imaging.
Risk Management
Safety depends on the dose, type, and how manganese dioxide gets into the body. Animal tests show low doses cause few problems, but high doses can hurt the brain. High levels may damage the choroid plexus and hippocampus, kill cells, and cause swelling. Some animals had memory and behavior problems after high exposure. Manganese dioxide nanoparticles can cross body barriers and build up in the brain. These effects depend on the amount and how the nanoparticles enter the body. Most human data comes from workers around manganese, who sometimes get symptoms like Parkinson’s disease. There is not much direct proof from medical use in people. To keep people safe, groups like the FDA ask for careful testing of manganese dioxide nanomaterials. Makers must check particle size, shape, and stability. They must show how these things link to safety and quality. Good manufacturing and stability tests help make safe products. The FDA also wants early checks for important quality features and unknown risks. These steps help protect patients and make sure manganese dioxide is safe in medicine.
Future Directions
Research Trends
Scientists are finding new ways to use manganese dioxide in medicine. Many are working on manganese dioxide nanosheets for drug delivery. These nanosheets can sense changes in the body, like pH or heat. Some even react to things outside the body, like light or magnets. Scientists want these systems to release drugs only when needed. This helps treatments work better and causes fewer side effects, especially for cancer.
Manganese dioxide nanosheets can act like enzymes. They also help with imaging by stopping fluorescence or turning on MRI signals. Some systems use all these features together for theranostic use. Scientists care about safety, so they use green methods and test for toxicity in living things. Targeting molecules, like aptamers and antibodies, help these systems find cancer cells.
New uses include photothermal therapy and chemo-dynamic therapy. Some scientists made manganese dioxide nanoparticles that can go into the brain. These help treat Alzheimer’s disease by changing the brain’s environment. Other studies use manganese dioxide in 3D-printed bone scaffolds or to boost the immune system. These examples show manganese dioxide is important in new medical technology.
Manganese dioxide systems react to body and outside signals.
Nanoplatforms can help with both therapy and imaging.
Safety and targeting are very important.
Clinical Challenges
It is hard to use manganese dioxide research in real medical care. The way manganese oxide nanoparticles act in the body and their toxicity matter a lot. Scientists must design new nanoparticles carefully to keep people safe and make them work well.
Nanotheranostic systems are complex, which makes them expensive and hard to approve. Developers must talk to rule-making groups to get products approved. There are many rules for drugs and devices, which makes things harder. Talking early and often with groups like the FDA and EMA helps get approval.
Many types of experts are needed. Teams need people who know physics, chemistry, engineering, biology, and medicine. It is hard to find good partners to make these products. If the dose, materials, or steps change, more safety tests are needed. This can slow down the work. New ways to test these products must be made and checked.
If a system has too many features, it can get too costly and not help patients more. Working with rule-makers early and teaming up with other groups helps lower risks and makes success more likely.
Challenge | Impact on Applications |
|---|---|
Limits safe use | |
Regulatory complexity | Slows approval |
Multidisciplinary demands | Requires broad expertise |
Manufacturing limitations | Delays clinical translation |
Analytical method gaps | Complicates validation |
Over-complex design | Raises cost, lowers benefit |
Tip: Working together early and having clear goals helps move manganese dioxide from the lab to the clinic.
Manganese dioxide nanomaterials help medicine move forward.
They make drug delivery better and help treat cancer.
Scientists use their special features for accurate tests and care.
New research checks if they are safe and can be used in hospitals.
Manganese dioxide will help create new medical tools and fight cancer.
FAQ
What makes manganese dioxide useful in drug delivery?
Manganese dioxide nanosheets can hold lots of medicine. They let the medicine out when the body has certain signals. This helps the medicine go to the right place. It also means fewer side effects for patients.
Is manganese dioxide safe for medical use?
Scientists check manganese dioxide to make sure it is safe. Most types are safe if the dose is low. High doses can be dangerous. Doctors and scientists watch how it is used very carefully.
Can manganese dioxide improve cancer treatment?
Manganese dioxide helps doctors treat cancer better. It adds more oxygen to tumors. It also helps medicine reach cancer cells. Doctors use it to make treatments stronger and more exact.
Related Posts

I am Edward lee, founder of manganesesupply( btlnewmaterial) , with more than 15 years experience in manganese products R&D and international sales, I helped more than 50+ corporates and am devoted to providing solutions to clients business.





