Manganese carbonate (MnCO₃) is used in many industries: feed additives, ceramics, water treatment, batteries, catalysts, and chemical synthesis. Because different applications require different levels of purity and performance, reading a manganese carbonate datasheet correctly is very important.
This guide explains every key indicator you will find in a manganese carbonate datasheet (also called COA – Certificate of Analysis). All data references are taken from ISO standards, GB industrial standards, EU Feed Additives Directive, and battery material technical references, so you can trust the accuracy.
A typical MnCO₃ datasheet includes:
| Parameter | Typical Value | Why It Matters |
|---|---|---|
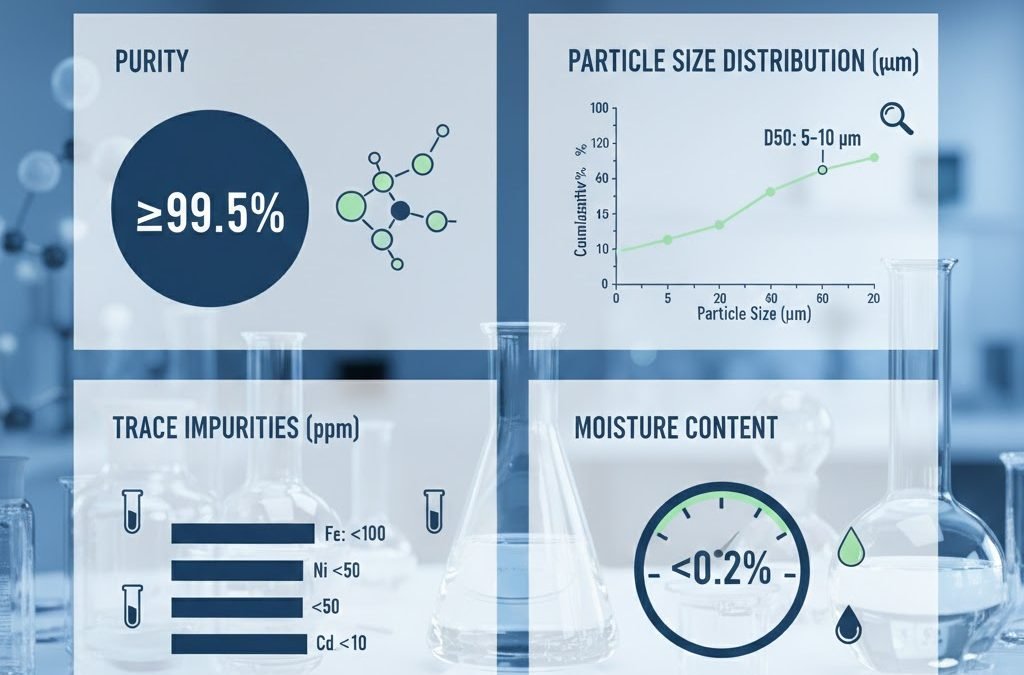
| MnCO₃ content (purity) | 90–99% | Determines usable active material |
| Mn content | 45–48% | Alternative purity indicator |
| Heavy metals (Pb, As, Cd, Hg) | <1–10 ppm | Regulated for feed and food industries |
| Fe impurity | 20–200 ppm | Affects color, battery performance |
| Cu, Zn, Ni | <5–20 ppm | Important for battery applications |
| Moisture | 0.1–1.0% | Controls stability and flowability |
| Particle size (D50) | 5–80 μm | Influences reactivity and dispersion |
| Bulk density | 0.5–1.4 g/cm³ | Affects packing and mixing behavior |
| Solubility | Required for feed grade | Important in animal nutrition |
| pH | 6–8 | Determines compatibility |
| Loss on ignition (LOI) | 30–34% | Confirms correct MnCO₃ chemistry |
These parameters follow several international standards:
✔ ISO 8289:2021 — Determination of manganese content
✔ EU Regulation 2002/32/EC — Heavy metal limits for feed additives
✔ GB/T 10500-2009 — Industrial manganese carbonate standard
✔ ASTM E1028 — Chemical analysis of manganese compounds
2. Purity (MnCO₃ content): The Most Important Indicator
Purity is the first line in every datasheet. It usually ranges:
Industrial grade: 90–94%
Feed grade: 94–98%
High-purity grade: 98–99%
Battery grade: >99%
According to GB/T 10500-2009, the minimum purity for high-grade manganese carbonate is ≥98%.
Why purity matters
In feed: low purity increases heavy-metal risks
In ceramics: impurities affect color
In batteries: trace impurities change charge efficiency
In catalysts: inconsistent reactions
Purity vs Mn content
MnCO₃ theoretical Mn content = 47.79% (calculated based on molecular weight).
So a sample with:
Mn = 47.5% → MnCO₃ ≈ 99.4%
Mn = 46.0% → MnCO₃ ≈ 96.3%
Source: molecular weight method from CRC Handbook of Chemistry and Physics, 2023.
3. Heavy Metals: Pb, As, Cd, Hg (Critical for Feed Grade)
Feed-grade manganese carbonate must comply with EU Regulation 2002/32/EC:
| Heavy Metal | EU Maximum Limit | Typical High-Quality MnCO₃ |
|---|---|---|
| Lead (Pb) | ≤10 ppm | 1–5 ppm |
| Arsenic (As) | ≤2 ppm | 0.3–1.5 ppm |
| Cadmium (Cd) | ≤1 ppm | ≤0.5 ppm |
| Mercury (Hg) | ≤0.1 ppm | ≤0.05 ppm |
If heavy metals exceed limits, the product cannot enter the EU feed market.
Why this matters:
Pb and As affect animal safety
Cd accumulates in organs
Heavy metals influence export customs clearance
4. Iron (Fe) Impurity: A Common Pain Point
Fe is the most common impurity in MnCO₃ because many mines contain iron. Fe levels vary widely:
Industrial-grade suppliers: 100–500 ppm
Good chemical-precipitation producers: 20–100 ppm
Why Fe matters:
Ceramics – Fe causes discoloration
Batteries – Fe reduces discharge efficiency
Catalysts – Fe interferes with reaction purity
Battery-grade MnCO₃ Fe limit :
Fe < 20 ppm
5. Particle Size Distribution (D50, D90)
Particle size changes how MnCO₃ behaves in your production environment.
| Application | Typical D50 | Why |
|---|---|---|
| Feed | 30–80 μm | Easy mixing, good digestibility |
| Ceramics | 1–10 μm | Higher reactivity, smoother finish |
| Batteries | 2–6 μm | Short diffusion pathways |
| Chemical synthesis | 10–50 μm | Controlled reaction rates |
D50 = the median particle size
D90 = 90% of particles are smaller than this value
Measured using laser diffraction (ISO 13320:2020).
6. Moisture: Controls Stability and Flowability
MnCO₃ should normally have moisture between 0.1% and 0.5%.
Why low moisture is important:
Moisture >1% causes caking
Moisture reduces effective MnCO₃ content
High moisture absorbs humidity during sea shipment
Industrial standard (GB/T 10500-2009):
Moisture ≤ 0.5%
7. Bulk Density (0.5–1.4 g/cm³)
Bulk density determines how the product flows in:
Feed mixers
Chemical reactors
Packaging systems
Low-density MnCO₃ = fluffy, easier dispersion
High-density MnCO₃ = better packing, less dust
Industry reference values:
Feed grade: 0.6–0.9 g/cm³
Battery grade: 1.0–1.3 g/cm³
8. Solubility (Feed Grade Requirement)
MnCO₃ itself is poorly soluble in water, but feed-grade MnCO₃ requires acid solubility >90% (in 0.1 M HCl).
This comes from AOAC Official Method 2016.03.
If solubility is low:
Animals cannot absorb Mn
Adds no nutritional value
High-quality feed MnCO₃ typically has acid solubility of 92–98%.
9. pH (Usually 6.0–8.0)
pH indicates impurity type:
pH <6 → too much acidic residue (sulfate/chloride)
pH >8.5 → excessive carbonate impurities
Safe range per industrial standard:
pH 6.0–8.0
10. LOI (Loss on Ignition): Confirms Correct Chemical Formula
MnCO₃ theoretical LOI = 34.4% (CO₂ release).
High-quality samples show 33–35% LOI.
If LOI is too low:
Contains MnO, impurities, or unreacted material
11. Example COA (Explained)
| Parameter | Result | Standard | Meaning |
|---|---|---|---|
| MnCO₃ | 99.1% | ≥99% | High purity |
| Mn | 47.5% | ≥47.0% | Matches purity |
| Pb | 1.8 ppm | <5 ppm | EU-feed safe |
| As | 0.9 ppm | <2 ppm | EU-feed safe |
| Fe | 35 ppm | <50 ppm | Suitable for ceramics / battery |
| Moisture | 0.12% | <0.5% | Very stable |
| D50 | 8 μm | 5–15 μm | Fine grade |
| pH | 7.2 | 6–8 | Chemically stable |
| LOI | 34.1% | 33–35% | Correct MnCO₃ |
This datasheet indicates a high-quality, high-purity product suitable for feed, ceramics, and battery use.
12. How to Identify a Reliable Supplier from a Datasheet
A good supplier provides:
Complete COA data (not only purity)
Heavy metal test reports (Pb, As, Cd, Hg)
Particle size analysis (ISO 13320)
Batch traceability
Third-party testing (SGS / Intertek)
Stable production process (chemical precipitation > mineral purification)
Signs of unreliable suppliers:
Purity but no heavy-metal data
No particle size data
Big variation between batches
Claims purity >99.5% (not realistic for MnCO₃)
13. Common Buyer Mistakes
❌ Only comparing price
❌ Ignoring impurities
❌ Not checking moisture
❌ Buying 90–92% purity for feed use (illegal for export)
❌ Not requesting COA before ordering
These mistakes can cause big financial loss.
14. Conclusion
Learning how to read a manganese carbonate datasheet helps you:
Avoid low-quality suppliers
Choose the correct grade for your application
Meet international regulations
Reduce production risks
Improve product consistency
As a professional MnCO₃ manufacturer, we can provide:
Industrial grade (90–94%)
Feed grade (94–98%)
High purity (98–99%)
Battery-grade MnCO₃ (>99%, low impurities)
Detailed COA, MSDS, and SGS test reports
FAQ
What purity level is recommended for feed-grade manganese carbonate?
Feed-grade manganese carbonate usually requires a purity of 94–98%, and it must comply with EU Regulation 2002/32/EC for heavy-metal limits. Higher-purity grades (98–99%) are used when stricter nutritional or regulatory requirements apply.Why is heavy-metal content (Pb, As, Cd, Hg) so important?
Heavy-metal levels determine whether the product is safe for feed, food-related applications, or export. For example, EU standards require Pb ≤10 ppm, As ≤2 ppm, Cd ≤1 ppm, and Hg ≤0.1 ppm. High heavy-metal content may cause customs issues, failed audits, or safety risks in animal production.How do I know if the manganese carbonate fits battery applications?
Battery-grade MnCO₃ must have >99% purity, Fe <20 ppm, and fine particle size (D50 around 2–6 µm). These specifications help improve charge efficiency and reduce impurities that can damage electrodes.What does D50 mean in a manganese carbonate datasheet?
D50 is the median particle size measured according to ISO 13320. It means 50% of particles are smaller than this size. Smaller D50 values improve reactivity and dispersion, which is important in ceramics, catalysts, and battery materials.Why is moisture content important when choosing manganese carbonate?
High moisture (>1%) can lead to caking, reduced purity, and problems during storage and shipment. Good-quality MnCO₃ typically contains 0.1–0.5% moisture, following GB/T 10500-2009 recommendations.What does LOI (Loss on Ignition) tell me?
LOI indicates how much CO₂ is released when MnCO₃ is heated. Pure manganese carbonate has a theoretical LOI of 34.4%. Values between 33–35% show correct MnCO₃ chemistry; lower values suggest impurities such as MnO or unreacted materials.How can I confirm if a supplier’s datasheet is reliable?
A reliable supplier provides complete COA data, ISO-based particle size analysis, stable heavy-metal results, and consistent batch records. Missing information—especially heavy-metal data—is usually a red flag for quality issues.What grade should I choose for ceramics?
Ceramics manufacturers typically use fine-particle MnCO₃ (D50 = 1–10 µm) and require low Fe impurity (<50 ppm). These specifications help maintain color stability and improve firing reactions.Why do Mn and MnCO₃ percentages differ on the datasheet?
MnCO₃ purity and Mn% are related but not identical. Theoretical Mn content in pure MnCO₃ is 47.79%, so Mn% can be used to calculate purity. For example, Mn = 47.5% ≈ MnCO₃ 99.4%.Do all applications require high-purity manganese carbonate?
No. Industrial-grade applications like pigments or generic catalytic reactions may only require 90–94% purity. Feed, battery, and electronic applications need higher grades due to stricter impurity and safety requirements.
Related Posts

I am Edward lee, founder of manganesesupply( btlnewmaterial) , with more than 15 years experience in manganese products R&D and international sales, I helped more than 50+ corporates and am devoted to providing solutions to clients business.