Electrolytic manganese dioxide is very important for batteries and water treatment. Companies use its high purity and special properties to make good batteries. These batteries are used in electric cars and for storing renewable energy. In 2024, people made about 300,000 tons of it worldwide. The amount keeps growing because more people in Asia-Pacific want it. Learning how this compound is made helps companies work better. It also helps them protect the environment and keep up with new technology needs.
Key Takeaways
- Electrolytic manganese dioxide (EMD) is very pure. It has a special structure. This helps batteries work better. It also helps them last longer. Most EMD is used in batteries. These batteries are for electric cars, phones, and energy storage. EMD is important for clean energy and technology. The production process uses natural ores. It also uses careful purification and electrolysis. This makes high-quality EMD. It also creates less waste and uses less energy. EMD helps clean water too. It removes pollutants quickly and safely. This is because of its strong chemical properties. Workers must wear safety gear when handling EMD. This keeps them healthy. Companies follow strict rules to protect the environment.
What Is Electrolytic Manganese Dioxide
Key Properties
Electrolytic manganese dioxide has a special crystal structure called δ-MnO2. This structure has layers and a big surface area. There are many empty spots for oxygen in it. These features help it store and give off energy fast. Water sits between the layers, which helps it conduct electricity better. EMD has more capacitance and less resistance than other manganese oxides. Its tiny particles and strong chemical activity help batteries work well. EMD lasts longer and works better as a depolarizer than natural manganese dioxide. It is very pure compared to other types. Most natural manganese dioxide has more impurities and less manganese. EMD is made by electrolysis, so it is pure enough for batteries.
Note: EMD’s high purity and special structure help it work better in tough jobs like batteries and water treatment.
Industrial Significance
Electrolytic manganese dioxide is important in many industries. Most EMD is used to make batteries. These batteries power electric cars, electronics, and energy storage. EMD also helps clean water by removing bad substances. It does this through oxidation and adsorption. Other industries use EMD too. These include steel, glass, chemicals, and electronics. They use it for its catalytic and redox properties.
| Industrial Sector | Estimated Market Share (2023) | Key Details |
|---|---|---|
| Battery Industry | 91.5% | Used in lithium-ion, alkaline, and zinc-carbon batteries |
| Water Treatment | N/A | Removes contaminants through oxidation and adsorption |
| Steel, Glass, Chemicals | N/A | Used for catalytic and redox reactions |
| Electronics | N/A | Supports electronic component manufacturing |
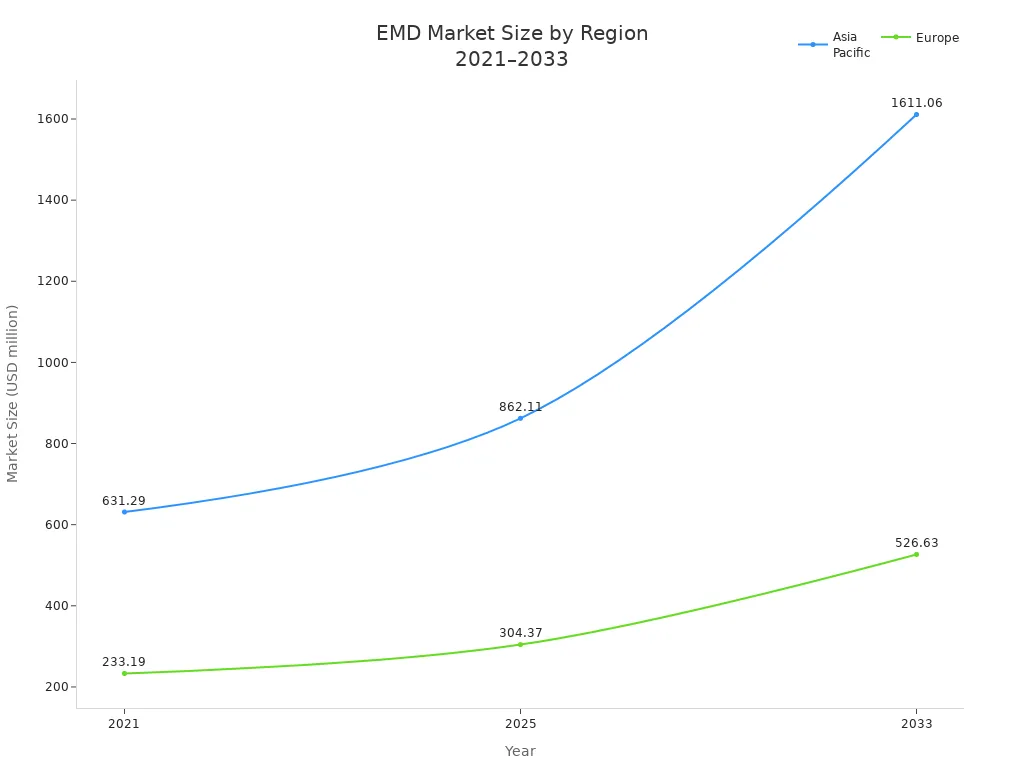
Asia-Pacific, mainly China, leads the EMD market. This is because it makes many batteries. North America and Europe also need a lot of EMD. This is due to electric cars and strict environmental rules.
Production Process
Raw Materials
Making electrolytic manganese dioxide starts with natural manganese ores. Pyrolusite is the main mineral used. It has about 60 to 63% manganese dioxide. Cryptomelane is another mineral that has potassium. Workers dig these ores from the ground. They clean the ores to take out things they do not want. Next, they use sulfuric acid to leach the cleaned ore. This makes a manganese sulfate solution. The solution is used for electrolysis.
- Main raw materials:
- Pyrolusite (high manganese dioxide content)
- Cryptomelane (contains potassium)
- Sulfuric acid (for leaching)
- Water (for solution preparation)
Note: Good ores and careful work help make a pure final product.
Purification Steps
Purification is very important for making good electrolytic manganese dioxide. First, workers mine the ore. Then, they crush and grind it into small pieces. They treat the ore to make manganese dissolve. They may use heat or magnets for this. After treatment, they leach the ore with sulfuric acid. This step dissolves the manganese. The solution goes through many cleaning steps. These steps remove iron, heavy metals, calcium, magnesium, sodium, and potassium. If these are not removed, batteries will not work well.
- Mine and crush manganese ore.
- Grind the ore to fine particles.
- Pre-treat by calcining or magnetic separation.
- Leach with sulfuric acid.
- Remove iron and heavy metals.
- Refine further by precipitating calcium and magnesium.
- Wash and filter to achieve high purity.
Each step removes certain things that should not be there. This makes sure the manganese sulfate solution is clean before electrolysis.
Electrolysis Method
Electrolysis is the main part of making electrolytic manganese dioxide. In this step, electricity goes through the clean manganese sulfate solution. Manganese dioxide forms on titanium anodes. This makes a very pure product. Workers watch many things to keep the quality high:
| Parameter | Typical Range | Effect on Product Quality |
|---|---|---|
| Current Density | 100 – 200 A/m² | Shapes deposit morphology and surface area |
| Voltage | 0.6 – 1.2 V | Lower voltages yield uniform, crack-free deposits |
| Temperature | 94°C – 120°C | Affects crystal size and surface area |
| Deposition Time | ~5 hours | Balances storage performance and porosity |
| MnSO4 Concentration | ~0.9 mole/liter | Maintains steady manganese supply |
| H2SO4 Concentration | ~0.5 mole/liter | Keeps electrolyte stable and acidic |
| Titanium Doping | Added as TiOSO4 | Improves structure and electrochemical performance |
They use titanium anodes and check the solution often. They take out hydrogen and add more solution when needed. This keeps everything working right. This method lets them control how pure and strong the manganese dioxide is. The product made this way is good enough for batteries.
Tip: Electrolysis gives better purity and is more reliable than chemical ways. That is why battery makers like it best.
Energy-Efficient Innovations
New ideas have made the process use less energy. Some new ways do not need the old heating step. This step used to need a lot of heat and energy. Skipping it saves energy and money. These changes also help the environment.
The electrolysis process has many good points:
- The product is very pure, which is great for batteries and electronics.
- Acid can be used again, so there is less waste.
- Fewer chemicals are needed, which saves money and helps nature.
The process costs a lot to set up, but the good results are worth it. The high quality and better care for the environment make it the best choice for important uses.
Electrolytic Manganese Dioxide Applications
Batteries
Electrolytic manganese dioxide is very important for batteries. Battery companies use it as a cathode in many battery types. These include alkaline, lithium-ion, sodium, and zinc–carbon batteries. These batteries power electric cars, phones, and energy storage. In 2023, over 91% of this material was used for batteries. Asia-Pacific makes and uses the most, but North America is growing too. Companies are making more to meet the need for electric cars and electronics.
This material helps batteries work better and last longer. In alkaline and zinc–carbon batteries, it gives high energy and stable use. Its structure lets zinc ions move in and out easily. This helps batteries charge and discharge many times. Some additives in the electrolyte can make a layer on the cathode. This layer helps the battery last longer. In lithium and sodium batteries, its high purity and special structure give more power and better charging.
Note: More people want this material for batteries. The market could reach $3 billion by 2030 because of electric cars and electronics.
Water Treatment
Electrolytic manganese dioxide is also used in water treatment. It acts as a catalyst to help clean water faster. It speeds up oxidation reactions to remove bad things from water. Its high tap density and redox properties make it work well. The small, even particles let ions move fast, so reactions happen quickly. Its redox behavior helps break down pollution.
Water plants use it to take out organic compounds and heavy metals. It works for cleaning both wastewater and drinking water. The material’s structure and surface defects help it react better. These features give more places for reactions to happen. Engineers check how fast it removes pollution and how much cleaner the water gets.
- Key benefits in water treatment:
- Removes organic pollution quickly
- Breaks down harmful chemicals well
- Works reliably because of its stable structure
Electronics
The electronics industry needs electrolytic manganese dioxide for batteries in many devices. These include phones, smartwatches, power tools, and home gadgets. Its high purity and steady quality help batteries last longer and work better. The market for this material in electronics is growing fast, especially in China and Japan.
Manufacturers use it mostly in alkaline and lithium-ion batteries for portable devices. Its high tap density and strong redox properties give steady power. These features also help make smaller and lighter devices. As more people use gadgets and electric cars, the need for this material will keep going up.
| Application Area | Key Role of EMD | Benefit |
|---|---|---|
| Batteries | Cathode material in alkaline, lithium, sodium, and zinc–carbon batteries | High energy, long life, stable cycling |
| Water Treatment | Catalyst for oxidation of pollutants | Fast, efficient removal of contaminants |
| Electronics | Battery component for portable devices | Reliable power, supports miniaturization |

Advantages and Safety
Purity and Performance
Manufacturers pick this material because it is very pure and works well. Its crystal structure has both β- and R-MnO2 forms. These forms make bigger tunnels for ions to move through. Bigger tunnels help the material store and give off energy better. Other types, like β-MnO2, have smaller tunnels. Smaller tunnels make it harder for ions to move. This lowers how well batteries work. The electrolysis process makes a smooth and thick coating. This way, there are fewer impurities and the shape is better. High purity helps batteries work better and last longer.
| Aspect | Details |
|---|---|
| Typical Purity Levels | Impurities less than 0.1 wt.% after purification steps |
| MnO2 Form | Mostly gamma-MnO2 (γ-MnO2), known for high electrochemical activity |
| Impact on Battery Performance | High purity gives strong electrochemical activity and stability, making batteries efficient and long-lasting |
| Battery Types Using EMD | Used in Leclanché cells, alkaline batteries, lithium-ion batteries |
| Electrolysis Process Advantages | Produces better properties, fewer impurities, regenerates acid, uses less reagent, eco-friendly |
Note: This material is the best choice for battery makers because it is pure and has a special structure.
Health and Environmental Notes
People must be careful when working with manganese dioxide. Workers in mines, smelting plants, and battery factories have the most risk. Breathing in manganese dust can cause health problems.
- Too much manganese in the body can hurt the brain and nerves. This can cause movement problems and changes in thinking.
- Breathing in the dust can make lungs sore, cause coughing, and lead to bronchitis.
- Being around manganese for a long time can cause brain diseases like Parkinson’s.
- Tiny manganese oxide particles can hurt cells by making harmful chemicals. This can kill cells or change their genes.
- Touching it can make skin itchy or cause allergies, but this does not happen often.
Employers must give workers safety gear and check the air for dust. Health checks help find problems early. Good safety and air flow keep workers and nature safe.
New changes have made EMD production much better. Companies now try to make EMD more pure and design smaller particles. They also want it to work better in batteries. Cleaner ways to make EMD and better ways to handle waste are now common.
- Since 2020, companies spent more money and improved their factories. This helped them make more EMD and move it faster.
- Scientists are working on ways to make EMD that are cheaper and better for the planet.
The EMD market will keep growing because more batteries are needed. New ideas like robots, tiny particle types, and new cleaning methods will help. These changes will support clean energy and new technology.
FAQ
What makes electrolytic manganese dioxide better than natural manganese dioxide?
Electrolytic manganese dioxide is much purer than natural manganese dioxide. Its crystal structure is also better for batteries. Battery makers like it because it helps batteries last longer and work better. Natural manganese dioxide has more things in it that are not wanted. It does not work as well in new types of batteries.
Is electrolytic manganese dioxide safe to handle?
Workers need to wear safety gear when working with EMD. Breathing in manganese dust can hurt the lungs and nerves. Companies give workers masks and good air flow to keep them safe. Health checks are done often to stop health problems before they get worse.
Which industries use the most electrolytic manganese dioxide?
Most EMD is used by the battery industry. Water treatment plants and electronics companies also use it. EMD is needed to make batteries for electric cars, phones, and storing energy.
How does EMD production affect the environment?
New ways to make EMD use less energy and make less waste. Recycling acid and using fewer chemicals help lower pollution. Companies now try to use cleaner methods to protect nature and follow strict rules.
Related Posts

I am Edward lee, founder of manganesesupply( btlnewmaterial) , with more than 15 years experience in manganese products R&D and international sales, I helped more than 50+ corporates and am devoted to providing solutions to clients business.





