Manganese dioxide (MnO₂) is a versatile inorganic compound widely used in industries ranging from battery manufacturing to pigments. Its unique chemical and physical characteristics make it particularly valuable in ceramics and glass production. This post explores its composition, roles, processing considerations, sourcing, and applications in detail, helping materials engineers, ceramic artists, and production managers optimize its use.
Chemical identity: MnO₂, also known as pyrolusite in its mineral form
Sources & production:
Naturally occurs in ores such as pyrolusite.
Commercially synthesized via chemical precipitation—commonly from potassium permanganate or manganese sulfate.
Physical characteristics:
Deep black or dark grey powder with a Mohs hardness of ~6–6.5 .
High melting point (~535 °C) and excellent thermal stability.
Industrial grades:
Ranges from ≥60% to ≥90% purity.
Tailored particle sizes and RoHS-compliant versions available for specific applications.
Role of Manganese Dioxide in the Ceramics Industry
1. Enhancing Ceramic Density
Ceramic bodies often face challenges during sintering—porosity, uneven densification, and weak mechanical structure. MnO₂ serves as an effective densifying agent:
During high-temperature firing (typically 1200–1400 °C), MnO₂ facilitates crystallization and promotes diffusion processes that reduce internal pores and microstructural defects.
These densification effects result in higher mechanical strength, improved durability, and enhanced resistance to cracking—crucial for industrial ceramics in aerospace, electronics, and mechanical components
2. Reducing Shrinkage in Firing
Ceramics shrink significantly during firing—often up to a third of initial volume.
MnO₂ helps stabilize the sintering process, reducing sudden shrinkage and maintaining dimensional integrity.
A stable shrinkage profile ensures consistent form, less warping and cracking, and lower scrap rates, which is essential in high-volume manufacturing lines.
3. Color Modulation in Glazes
Beyond mechanical benefits, MnO₂ provides artistic value in glaze coloration:
Even small percentages produce rich, earthy tones—warm browns, deep greens, amber hues. This emerges from complex interactions with other oxides (e.g., Fe, Ti, Ca) during firing.
MnO₂ improves gloss and opacity, giving glazes a smooth, lustrous finish highly prized by potters and tile manufacturers.
4. Boosting Glaze Hardness and Durability
MnO₂ contributes to functional performance, not just aesthetics:
Glazes infused with MnO₂ exhibit enhanced surface hardness and improved scratch resistance—ideal for tableware and high-traffic tiles.
They also demonstrate stronger chemical resistance, tolerating acidic and alkaline exposure, making them well-suited for labware or outdoor architectural ceramics.
| Feature | Effect of MnO₂ in Ceramics |
|---|---|
| Density & strength | Promotes crystallization, reduces pores |
| Dimensional stability | Minimizes shrinkage and cracking |
| Glaze aesthetics | Yields rich, earthy hues and increased opacity |
| Surface performance | Enhances hardness and chemical resistance |

Manganese Dioxide in Glass Manufacturing
A. A Master of Color Control
MnO₂ serves dual roles:
At higher concentrations: acts as a colorant, producing vivid purple, blue, and black tones—effectively used by ancient artisans and modern glassmakers .
Example: Roman violet glass, prized for its deep purple tints, contained high MnO₂ levels.
B. The Science of Decolorization
Ferrous iron (Fe²⁺) impurities in silica raw materials cause a green tint.
MnO₂ oxidizes Fe²⁺ to Fe³⁺ (which is yellowish), while MnO₂ itself partially forms Mn³⁺ (purple).
Their complementary colors (yellow + purple) neutralize the green hue, resulting in crystal-clear glass—essential for beverage bottles, windowpanes, and optical components.
Modern glass manufacturing depends on this interplay to produce high-clarity transparent glass adequate for food, pharmaceutical, and decorative uses.
C. Industrial and Artistic Synergy
Architectural glass: subtle tints for UV protection and aesthetic appeal.
Decorative craftwork: stained glass and artistic sculptures benefit from MnO₂ coloration.
Functional specialty glass: laboratory ware and solar glass utilize MnO₂ for its stability under heat and chemicals.
In summary, MnO₂ enables both precise color control and functional transparency, bridging technical and creative needs.

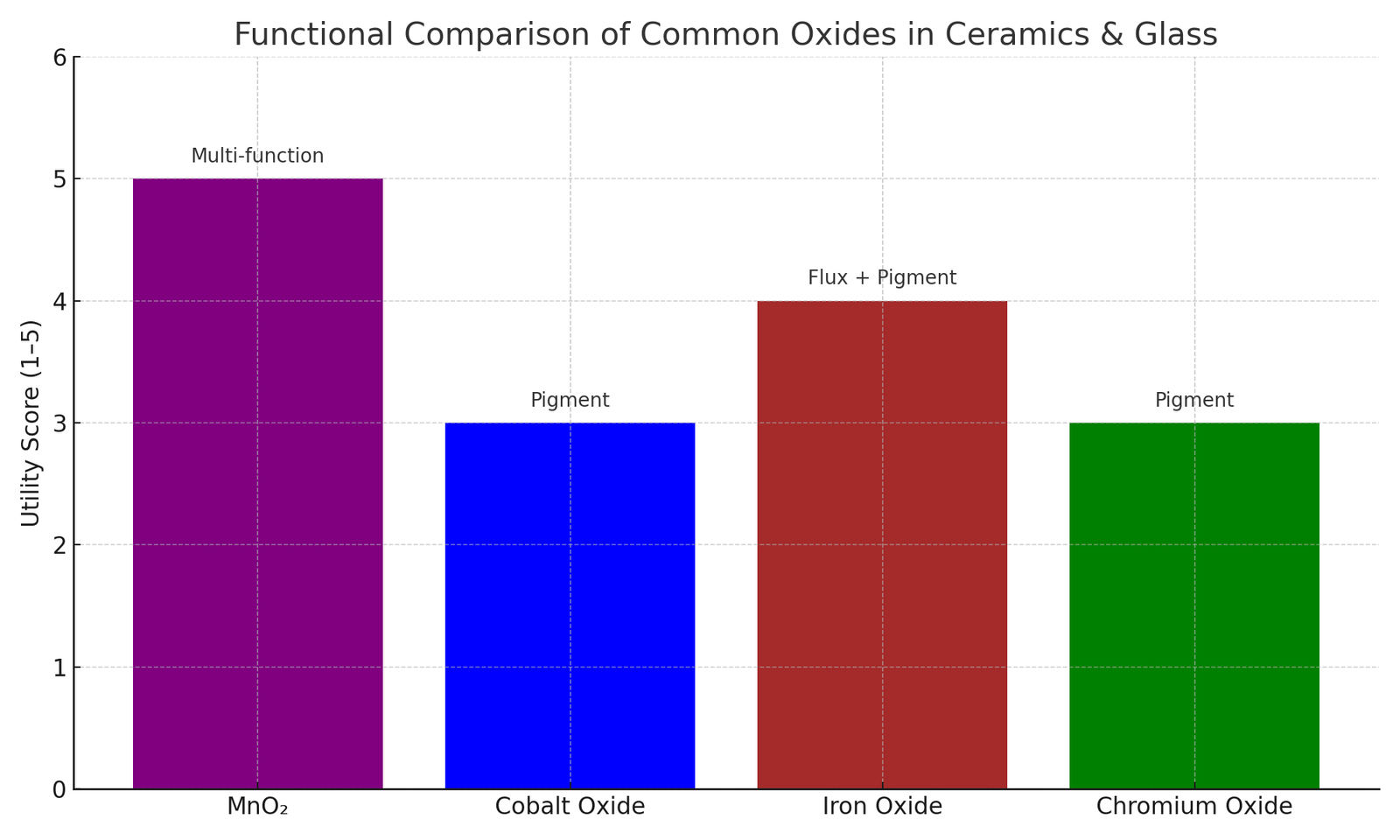
Comparison with Other Oxides in Ceramics & Glass
| Oxide | Primary Function | Color or Effect |
|---|---|---|
| Manganese Dioxide | Colorant, decolorizer, densifier | Purple, amber, black, densification aid |
| Cobalt Oxide | Pigment | Deep blue |
| Iron Oxide | Colorant, flux | Red, brown, green |
| Chromium Oxide | Colorant | Green |
MnO₂ stands out due to its multifunctionality—from structural benefits in ceramics to color and clarity control in glass.
Technical Considerations
Dosage Guidelines
Ceramics: typically 0.5–5 wt% for glazes and sintering aid.
Glass: 1–10 ppm for decolorizing; 0.1–1 wt% for coloring.
Compatibility
Mixes well with common oxides like silica, alumina, and calcium.
Interacts synergistically with iron, cobalt, and titanium oxides to enhance color dynamics.
Thermal Profile
To leverage sintering benefits, fire to 1200–1400 °C in oxidizing atmosphere.
In glass, oxidation control is crucial—reducing conditions can lead to darkened or opaque results.
Safety & Environmental
Wear dust protection; fine MnO₂ is irritating if inhaled.
Waste disposal must meet heavy metal regulations.
Prefer RoHS-certified grades for compliance.
Sourcing Manganese Dioxide for Ceramics & Glass
Key specifications:
Purity: ≥60%, ≥70%, or ≥90% thresholds depending on application.
Particle size: fine powders (e.g., D50 <10 µm) for glaze integration; coarser grades for sintering bodies.
Compliance: RoHS, EPA limits on lead/cadmium, etc.
Stability & packaging: moisture-resistant, inert packaging to preserve quality.
ManganeseSupply is a professional manufacturer and global supplier of manganese dioxide products with decades of experience serving the ceramics and glass industries.
Why Choose ManganeseSupply?
| Feature | Details |
|---|---|
| Product Purity | MnO₂ content ≥ 60%, ≥ 70%, or ≥ 90% based on your formulation needs |
| Custom Specification | Particle size control, moisture content <1%, RoHS-compliant |
| Application Expertise | Specific grades for glass decolorization, ceramic sintering, and glazes |
| Reliable Delivery | Global shipping with stable lead times |
| Technical Support | SDS, COA, and test data available upon request |
You can check out our full glass and ceramics solutions and download tds and msds for the following applications:
Glass Decolorizing Agent – Offsets iron-induced green tint in soda-lime glass production.
Neutralizing Green Tints in Glass – Improves clarity in container and flat glass manufacturing.
Optical Glass Manufacturing – High purity MnO₂ for controlled light transmission and minimal impurities.
Heat-Absorbing Architectural Glass – Regulates solar absorption and color tone stability.
Dark Brown & Black Ceramic Glazes – Produces deep, stable glaze coloration during firing.
High-Temperature Porcelain Glazes – Maintains color integrity in high firing ranges.
Enamel and Frits Industry (90% Grade) – Provides oxidation control and consistent pigment interaction.
Ceramic Body Stains and Pigments – Enhances brown/black pigment formulation stability.
Clay Brick Coloring (Natural Pyrolusite Powder) – Cost-effective mineral coloring agent.
Artisan Glass Blowing Studios – Concentrated MnO₂ for small-batch glass coloration.
Conclusion
MnO₂ offers unique advantages across ceramics and glass:
Structural strength & density in engineered ceramics.
Dimensional control and reduced waste during firing.
Aesthetic depth in glazes via rich earthy tones.
Surface resilience, with increased hardness and resistance.
Precision color control in glass, from decolorization to intense hues.
Selecting the right grade, dosage, and firing profile ensures these benefits manifest in quality results. Contacting reputable suppliers is the first step toward leveraging MnO₂’s full potential.
FAQs
Can MnO₂ be used with cobalt or iron oxides?
Yes—combining these oxides enhances color complexity. For example, cobalt + MnO₂ can yield rich greens; iron + MnO₂ deepens brown shades.
What firing temperature is ideal when using MnO₂?
For ceramics, 1200–1400 °C allows adequate sintering and glaze reaction. In glass, melting is done between 1400–1550 °C with controlled oxidation.
Is MnO₂ safe for food-contact ware?
When fully vitrified in glaze (no leaching), MnO₂ is food-safe. It’s common in dinnerware. Ensure glazes pass certifications like FDA or EN 1183.
Which grade is best for decolorizing glass?
Ultra-pure MnO₂ (≥90%) with small particle size ensures efficient oxidation of Fe²⁺ without introducing haze or residual color.

I am Edward lee, founder of manganesesupply( btlnewmaterial) , with more than 15 years experience in manganese products R&D and international sales, I helped more than 50+ corporates and am devoted to providing solutions to clients business.





