You can learn how manganese sulfate is made by mixing manganese dioxide with sulfuric acid or sulfur dioxide. The process involves easy materials and simple steps. The table below illustrates common reactions:
Reactants | Reaction | Product |
|---|---|---|
MnO₂ + H₂SO₄ → MnSO₄ | Manganese sulfate | |
Manganese dioxide + Sulfur dioxide | MnO₂ + SO₂ → MnSO₄ | Manganese sulfate |
Always remember to think about safety and purity when you use chemicals.
Manganese sulfate is made by mixing manganese dioxide with sulfuric acid or sulfur dioxide. Pick the way to make it based on what materials you have.
Safety is very important when you work with chemicals. Always wear safety gear and make sure your workspace has fresh air.
Purifying manganese sulfate is needed for good quality uses. Use ion exchange and adsorption to take out things that should not be there.
How Manganese Sulfate is Made
Materials
You need the right materials to begin. The main raw materials for how manganese sulfate is made are manganese oxide, sulfuric acid, and sometimes manganese carbonate. These materials react together to make the manganese sulfate solution you want.
Raw Material | Description |
|---|---|
Manganese Oxide | Main material for making manganese sulfate |
Sulfuric Acid | Important acid for the reaction |
Manganese Carbonate | Another material you can use |
You also need equipment to make manganese sulfate safely. Here are some common tools:
Stirrer: Mixes everything well. You need at least 5 kW for each cubic meter of slurry.
Sulfur Dioxide Gas: Used in some ways for leaching.
Oxygen: Sometimes added to help the reaction work.
Filtration tools: Separate solids from the manganese sulfate solution.
Crystallization dishes or tanks: Collect the final product.
Some advanced methods need extra equipment:
Precipitation system: Uses pH changes and filtration.
Solvent extraction tools: Help separate metal ions very well.
Safety Tips
You must always think about safety when working with chemicals in how manganese sulfate is made. Manganese dioxide, sulfuric acid, and sulfur dioxide can be dangerous if not handled right.
Use goggles and keep an eye wash station nearby.
Use approved respirators if there is dust or fumes.
Store sulfuric acid in a cool, dry place in sealed containers.
Install sulfur dioxide detectors and check them often.
Make sure the room has good airflow to remove sulfur dioxide gas.
Train everyone on what to do in emergencies, like evacuation and first aid.
Always have an eye-wash kit ready for spills.
Tip: Never work alone with strong acids or gases. Always tell someone when you use these chemicals.
Main Methods
There are several main ways to make manganese sulfate. Each way uses different materials and steps, but all try to make pure manganese sulfate solution.
Manganese Dioxide with Sulfuric Acid
Mix manganese dioxide with sulfuric acid. The reaction makes manganese sulfate and water. This way is simple and used in labs.Manganese Dioxide with Sulfur Dioxide and Water
Pass sulfur dioxide gas through manganese dioxide and water. This makes manganese sulfate. You need special tools to handle the gas safely.Leaching Manganese Ore
Use manganese ore as the starting material. Add sulfuric acid and sometimes sulfur dioxide. The ore dissolves and you get a manganese sulfate solution. This way is popular in factories because it uses natural ore.Manganese Metal with Sulfuric Acid
Dissolve pure manganese metal in sulfuric acid. This gives you very pure manganese sulfate solution. It needs less cleaning than ore-based ways.
The two most common ways in factories are leaching manganese ore and dissolving manganese metal in sulfuric acid. If you use ore, you must leach, clean, and crystallize the product. If you use manganese metal, you can skip some steps and get higher purity. The choice depends on what materials you have and the quality you want.
Method | Environmental Impact |
|---|---|
Leaching | Can cause pollution from manganese ions, make wastewater, and raise ion levels. |
Solidification | Good at trapping Mn2+, but may leak after long storage. |
You should always think about the environment with each process. Leaching can make wastewater with lots of manganese. Solidification traps manganese ions but might leak later.
Note: Always follow local rules for waste and protecting the environment when making manganese sulfate.
Production Process
Prepare the Reaction
First, get your materials and tools ready. Pick your method based on what you have. If you use manganese ore and sulfuric acid, measure them carefully. If you use manganese metal, use pure metal and sulfuric acid. Make sure your reactor and stirrer are clean. Put on gloves, goggles, and a respirator. The room should have good airflow. Prepare chemicals where air moves well. Always keep an eye wash station close by.
You can watch the reaction with special tools. NMR spectroscopy shows how fast the reaction goes. Some labs use PTFE tubing or glass flow cells to check all the time. You can also take small samples every hour. Use X-ray diffraction to see how much manganese sulfate forms. This lets you check the process without stopping it.
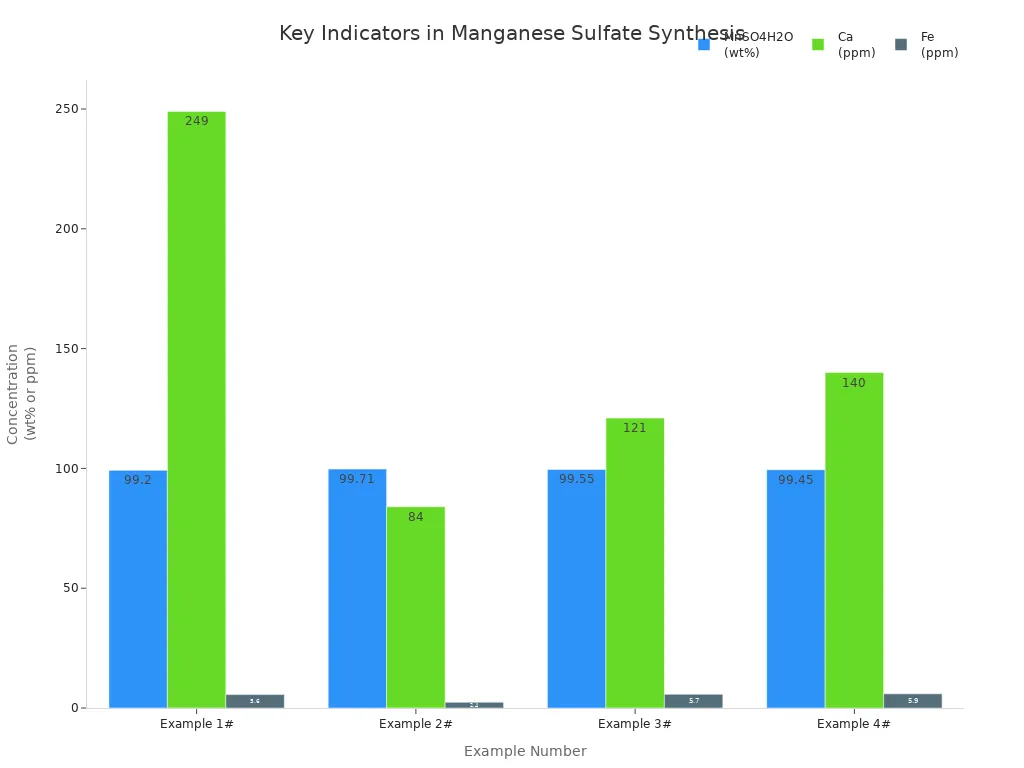
A good reaction gives a solution with lots of manganese and few impurities. The table below shows what you want in your product:
Indicator | Good Value |
|---|---|
MnSO4H2O (wt%) | 99.2–99.7 |
K, Na, Mg, Fe | <50 ppm |
Ca | 84–249 ppm |
Pb, Cd, Co | Not detected |
Make Manganese Sulfate: Step-by-Step
You can make manganese sulfate in different ways. Here are two common ways:
Using Manganese Ore and Sulfuric Acid
Get manganese ore or dust with manganese oxides and carbonates.
Add sulfuric acid to the ore. Use 0.5 to 3 times the manganese amount.
Add a reductant like oxalic acid or coke. Use 0.1 to 2 times the manganese amount.
Stir the mix and let it react. Manganese comes out as manganese sulfate.
Collect the first liquid, which has manganese sulfate.
Add calcium hydroxide to the liquid. This step removes bad stuff and makes it cleaner.
Using Manganese Metal and Sulfuric Acid
Put manganese metal and sulfuric acid in your reactor.
The reaction makes manganese sulfate and hydrogen gas.
The reaction is:
Mn + H2SO4 → MnSO4 + H2Collect the liquid after the bubbles stop.
For both ways, keep the temperature and time right. The reaction often needs about 250°C for 120 minutes. Leaching works well at 95°C for 60 minutes.
Tip: Watch out for hydrogen gas when using manganese metal. Work where air moves well and keep flames away.
Purification
After making manganese sulfate, you need to clean it. This step is important for battery or high-purity uses. The solution may have things like calcium, magnesium, potassium, sodium, selenium, arsenic, and lead.
Remove iron by adding CaCO3 and stirring. This can take out almost all the iron.
Use air or oxygen and add CaCO3 at pH above 5.8. This helps remove iron and other metals.
Use hydrogen peroxide to help get more manganese and remove some bad stuff.
Take out potassium using the granite phase. Remove arsenic and lead by turning them into sulfides.
For very pure manganese sulfate, use ion exchange cleaning. Chelating resins like Lewatit® MDS TP220 work well for heavy metals.
Adsorption with manganese oxides also helps clean the solution.
High-purity manganese sulfate is needed for good lithium battery materials. Heavy metals can make the quality worse, so you must remove them.
You can check how pure your solution is with UV-Vis spectroscopy or ICP-OES. These tests tell you how much manganese and other metals are in your solution.
Crystallization
Finish by turning the clean manganese sulfate into crystals. This step makes your solution into solid crystals.
Technique | Temperature Range | Crystal Form |
|---|---|---|
Formation of pentahydrate | Below ~26°C | Pentahydrate |
Formation of tetrahydrate | Above 26°C | Tetrahydrate |
Slow evaporation method | 26–29°C | Tetrahydrate |
Let the solution cool slowly or let it evaporate slowly. If you keep it below 26°C, you get pentahydrate crystals. If you keep it above 26°C, you get tetrahydrate crystals. Both are good, but pick what you need.
Good manganese sulfate crystals look pink or white and are powdery. They melt at about 700°C and boil at 330°C.
Use airtight containers to keep out water.
Always seal the package to stop dirt from getting in.
⚠️ Safety Alert: Do not eat, drink, or smoke when handling manganese sulfate. Wear gloves, goggles, and protective clothes. Wash your hands after touching it. Do not breathe dust or fumes. Store it safely and follow all safety rules.
If you follow these steps, you can make manganese sulfate with high yield and purity. Lab production often gets more than 95% extraction. The final manganese sulfate meets world standards and works well for batteries and other uses.
You can make good manganese sulfate if you follow each step. Use safe ways to make it. Always watch the temperature. Check the pH often. Make sure the reaction does not go too long or too short.
Description | |
|---|---|
Control Temperature | This keeps the product stable while you make it. |
Monitor pH | This helps keep the right chemical setting. |
Regulate Reaction Time | This makes sure you get the quality you want. |
Look at your manganese sulfate crystals. They should be pink or white. Store them in a safe place. To make more, learn about the reaction steps. Always keep safety and quality in mind.
FAQ
How can you test the purity of manganese sulfate?
You can use UV-Vis spectroscopy or ICP-OES. These tests show how much manganese and other metals are in your sample.
What color should pure manganese sulfate crystals be?
Pure manganese sulfate crystals look pink or white. You should see a powdery texture.
Tip: Store crystals in a dry, cool place.
Is manganese sulfate safe to handle at home?
You must wear gloves, goggles, and a mask. Always work in a well-ventilated area.
Safety Gear | Purpose |
|---|---|
Gloves | Protect skin |
Goggles | Shield eyes |
Mask | Block dust/fumes |
Related Posts

I am Edward lee, founder of manganesesupply( btlnewmaterial) , with more than 15 years experience in manganese products R&D and international sales, I helped more than 50+ corporates and am devoted to providing solutions to clients business.





